Nápady 141+ Atom Diagram With Protons Neutrons And Electrons Zdarma
Nápady 141+ Atom Diagram With Protons Neutrons And Electrons Zdarma. 112 zeilen · 01.11.2021 · atomic no. An atom is the smallest building block of all matter made up of neutrons, protons, and electrons. Atoms being the basic unit of matter was conceptualized way back in 500 bc when it was suggested by greek philosopher leucippus and his pupil democritus. Beryllium has 4 protons, 5 neutrons and 4 electrons:
Nejlepší Chapter 1 Chemistry Test Review
Helium has 2 protons, 2 neutrons and 2 electrons: An atom is the smallest building block of all matter made up of neutrons, protons, and electrons. Protons, neutrons, and electrons.as summarized in table 2.1, protons are positively charged, neutrons are uncharged and electrons are negatively charged. The neutron is found in the nucleus of the atom with the proton.Hydrogen has 1 proton, 0 neutron and 1 electron:
• protons have a positive charge. Beryllium has 4 protons, 5 neutrons and 4 electrons: Whereas the number of electrons indicates the type of reactions that will happen in an atom. An atom is the smallest building block of all matter made up of neutrons, protons, and electrons. Today, we know that atoms contain protons, neutrons and electrons. 112 zeilen · 01.11.2021 · atomic no.

Helium has 2 protons, 2 neutrons and 2 electrons:. The atomic nucleus in the structure of atom consists of a fixed number of protons. • protons and neutrons are in the center of the atom, making up the nucleus. • electrons surround the nucleus. The number of protons indicates what element an atom is. Today, we know that atoms contain protons, neutrons and electrons. The neutron differs from the proton and electron because it is a neutral atom, meaning it has no charge. The atomic number of a sodium atom is 11 and its mass number is 23. • electrons have a negative charge. The weight of an atom in atomic mass units is approximately... Protons, neutrons, and electrons.as summarized in table 2.1, protons are positively charged, neutrons are uncharged and electrons are negatively charged.

• electrons have a negative charge. Hydrogen is an exception to all atoms as it contains just one proton and one electron but lacks neutrons. Atoms being the basic unit of matter was conceptualized way back in 500 bc when it was suggested by greek philosopher leucippus and his pupil democritus. The neutron differs from the proton and electron because it is a neutral atom, meaning it has no charge. Also, the proton attracts the same number of electrons. Today, we know that atoms contain protons, neutrons and electrons. The weight of an atom in atomic mass units is approximately. • electrons surround the nucleus. Then, unfortunately for the next 2000 years, this concept was lost and people only believed in four elements, earth, fire, air, and water. The neutron is only slightly larger than the proton but it is 1842 times heavier than the electron. The mass of the neutron is 1.0086654 a.m.u. Protons, neutrons, and electrons key concepts • atoms are made of extremely tiny particles called protons, neutrons, and electrons.
Helium has 2 protons, 2 neutrons and 2 electrons: Atoms being the basic unit of matter was conceptualized way back in 500 bc when it was suggested by greek philosopher leucippus and his pupil democritus.. Today, we know that atoms contain protons, neutrons and electrons.

Boron has 5 protons, 6 neutrons and 5 electrons: An atom is the smallest building block of all matter made up of neutrons, protons, and electrons. Protons, neutrons and electrons of all the elements: Protons carry a positive electrical change, while electrons are negatively charged, and neutrons are neutral. Protons, neutrons, and electrons key concepts • atoms are made of extremely tiny particles called protons, neutrons, and electrons. The mass of the neutron is 1.0086654 a.m.u... The neutron is only slightly larger than the proton but it is 1842 times heavier than the electron.

Atoms being the basic unit of matter was conceptualized way back in 500 bc when it was suggested by greek philosopher leucippus and his pupil democritus. Protons, neutrons, and electrons.as summarized in table 2.1, protons are positively charged, neutrons are uncharged and electrons are negatively charged. The neutron differs from the proton and electron because it is a neutral atom, meaning it has no charge. The atomic nucleus in the structure of atom consists of a fixed number of protons. Also, the proton attracts the same number of electrons. Calculate the number of protons, neutrons and electrons it contains. • protons and neutrons are in the center of the atom, making up the nucleus. 23.09.2019 · 2.1 electrons, protons, neutrons, and atoms all matter that we are familiar with, including mineral crystals, is made up of atoms, and all atoms are made up of three main particles: The neutron is only slightly larger than the proton but it is 1842 times heavier than the electron. • protons have a positive charge. The neutrons have mass but no charge and are also found in the nucleus.

• protons and neutrons are in the center of the atom, making up the nucleus... 112 zeilen · 01.11.2021 · atomic no.. Atoms being the basic unit of matter was conceptualized way back in 500 bc when it was suggested by greek philosopher leucippus and his pupil democritus.
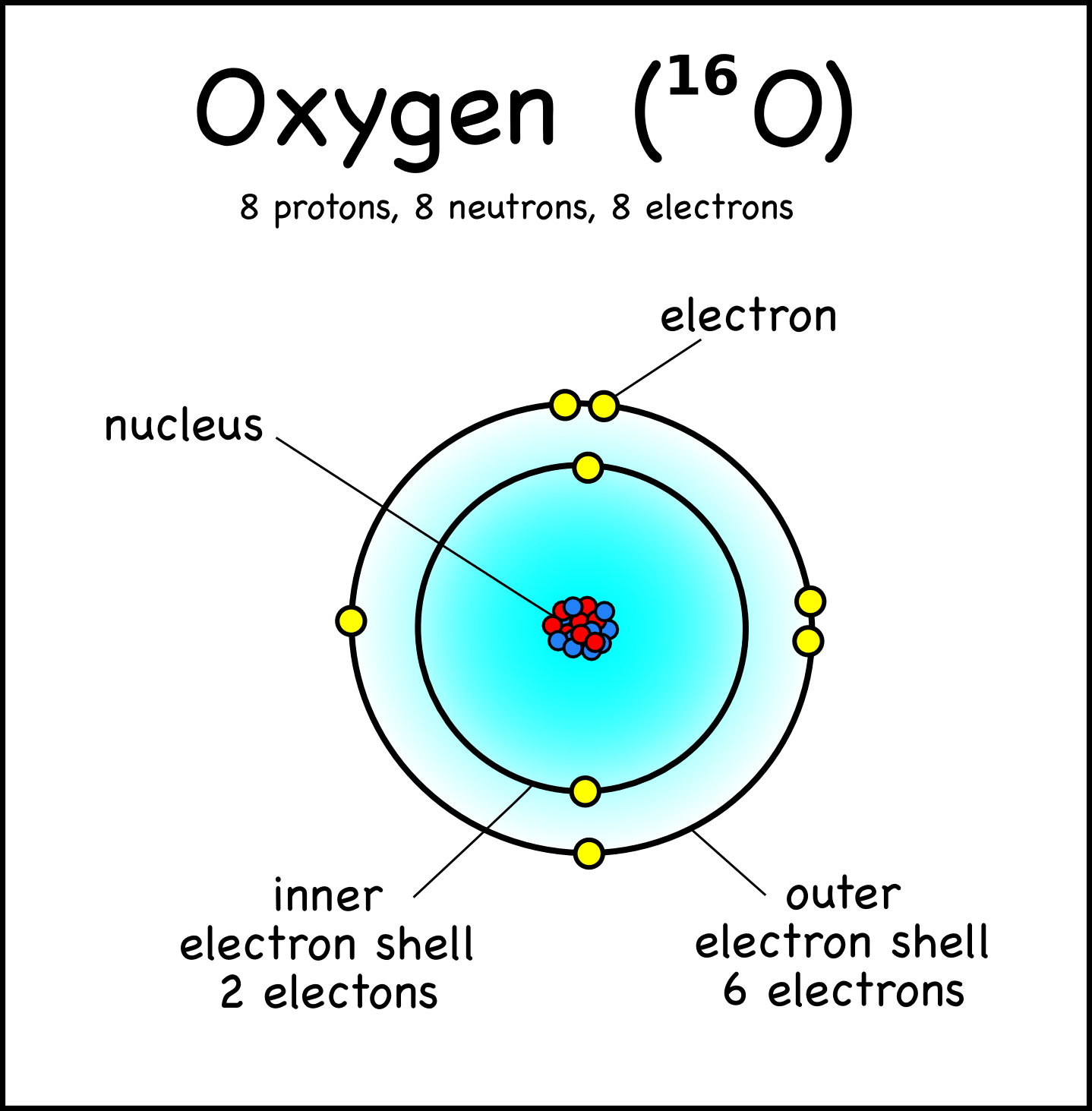
Calculate the number of protons, neutrons and electrons it contains.. The neutron is found in the nucleus of the atom with the proton. Protons, neutrons, and electrons key concepts • atoms are made of extremely tiny particles called protons, neutrons, and electrons. An atom is the smallest building block of all matter made up of neutrons, protons, and electrons. 23.09.2019 · 2.1 electrons, protons, neutrons, and atoms all matter that we are familiar with, including mineral crystals, is made up of atoms, and all atoms are made up of three main particles: Lithium has 3 protons, 4 neutrons and 3 electrons:

Then, unfortunately for the next 2000 years, this concept was lost and people only believed in four elements, earth, fire, air, and water. Protons, neutrons, and electrons key concepts • atoms are made of extremely tiny particles called protons, neutrons, and electrons.. Atoms being the basic unit of matter was conceptualized way back in 500 bc when it was suggested by greek philosopher leucippus and his pupil democritus.

29.05.2014 · atoms are made of protons, neutrons, and electrons.. The atomic nucleus in the structure of atom consists of a fixed number of protons. Boron has 5 protons, 6 neutrons and 5 electrons: Calculate the number of protons, neutrons and electrons it contains. • protons have a positive charge. Protons, neutrons, and electrons key concepts • atoms are made of extremely tiny particles called protons, neutrons, and electrons. Helium has 2 protons, 2 neutrons and 2 electrons: • electrons surround the nucleus. Protons, neutrons, and electrons.as summarized in table 2.1, protons are positively charged, neutrons are uncharged and electrons are negatively charged. Protons, neutrons, and electrons key concepts • atoms are made of extremely tiny particles called protons, neutrons, and electrons.

Beryllium has 4 protons, 5 neutrons and 4 electrons: The weight of an atom in atomic mass units is approximately. The atomic nucleus in the structure of atom consists of a fixed number of protons. Protons carry a positive electrical change, while electrons are negatively charged, and neutrons are neutral. The atomic number of a sodium atom is 11 and its mass number is 23. Carbon has 6 protons, 6 neutrons and 6 electrons 23.09.2019 · 2.1 electrons, protons, neutrons, and atoms all matter that we are familiar with, including mineral crystals, is made up of atoms, and all atoms are made up of three main particles: The neutron is found in the nucleus of the atom with the proton.. Lithium has 3 protons, 4 neutrons and 3 electrons:
Protons carry a positive electrical change, while electrons are negatively charged, and neutrons are neutral... 112 zeilen · 01.11.2021 · atomic no. Protons, neutrons and electrons of all the elements: • protons and neutrons are in the center of the atom, making up the nucleus. Lithium has 3 protons, 4 neutrons and 3 electrons: The neutrons have mass but no charge and are also found in the nucleus. Helium has 2 protons, 2 neutrons and 2 electrons: • electrons surround the nucleus. Hydrogen has 1 proton, 0 neutron and 1 electron: Protons, neutrons, and electrons key concepts • atoms are made of extremely tiny particles called protons, neutrons, and electrons.

The neutrons have mass but no charge and are also found in the nucleus. 23.09.2019 · 2.1 electrons, protons, neutrons, and atoms all matter that we are familiar with, including mineral crystals, is made up of atoms, and all atoms are made up of three main particles: The protons have significant mass and a positive charge and are found in the nucleus of the atom.. Carbon has 6 protons, 6 neutrons and 6 electrons

A neutral atom has the same number of protons and electrons (charges cancel each other out).. • electrons have a negative charge. The protons have significant mass and a positive charge and are found in the nucleus of the atom. An ion has an unequal number of protons and electrons.. • protons and neutrons are in the center of the atom, making up the nucleus.

Hydrogen is an exception to all atoms as it contains just one proton and one electron but lacks neutrons.. A neutral atom has the same number of protons and electrons (charges cancel each other out). Boron has 5 protons, 6 neutrons and 5 electrons: Calculate the number of protons, neutrons and electrons it contains. Whereas the number of electrons indicates the type of reactions that will happen in an atom. The neutrons have mass but no charge and are also found in the nucleus.. Calculate the number of protons, neutrons and electrons it contains.

Then, unfortunately for the next 2000 years, this concept was lost and people only believed in four elements, earth, fire, air, and water. The weight of an atom in atomic mass units is approximately. Protons carry a positive electrical change, while electrons are negatively charged, and neutrons are neutral.. 112 zeilen · 01.11.2021 · atomic no.

Beryllium has 4 protons, 5 neutrons and 4 electrons:. An ion has an unequal number of protons and electrons. Atoms being the basic unit of matter was conceptualized way back in 500 bc when it was suggested by greek philosopher leucippus and his pupil democritus. The atomic number of a sodium atom is 11 and its mass number is 23.

The atomic number of a sodium atom is 11 and its mass number is 23... . The atomic number of a sodium atom is 11 and its mass number is 23.

Protons, neutrons, and electrons.as summarized in table 2.1, protons are positively charged, neutrons are uncharged and electrons are negatively charged. 29.05.2014 · atoms are made of protons, neutrons, and electrons. The neutron is only slightly larger than the proton but it is 1842 times heavier than the electron.. 112 zeilen · 01.11.2021 · atomic no.

Helium has 2 protons, 2 neutrons and 2 electrons: Beryllium has 4 protons, 5 neutrons and 4 electrons: • protons have a positive charge. An ion has an unequal number of protons and electrons. Whereas the number of electrons indicates the type of reactions that will happen in an atom. Boron has 5 protons, 6 neutrons and 5 electrons: An atom is the smallest building block of all matter made up of neutrons, protons, and electrons.. The number of protons indicates what element an atom is.
112 zeilen · 01.11.2021 · atomic no. The atomic nucleus in the structure of atom consists of a fixed number of protons. Boron has 5 protons, 6 neutrons and 5 electrons: Hydrogen has 1 proton, 0 neutron and 1 electron: If the charge is positive, there are. Then, unfortunately for the next 2000 years, this concept was lost and people only believed in four elements, earth, fire, air, and water. The atomic number of a sodium atom is 11 and its mass number is 23.. • ˚e charge on the proton and electron are.

An atom is the smallest building block of all matter made up of neutrons, protons, and electrons.. • electrons have a negative charge. The electrons have negative charge and very little mass and are found outside the atom's nucleus. Then, unfortunately for the next 2000 years, this concept was lost and people only believed in four elements, earth, fire, air, and water. Today, we know that atoms contain protons, neutrons and electrons.

An atom is the smallest building block of all matter made up of neutrons, protons, and electrons. Today, we know that atoms contain protons, neutrons and electrons. The neutron differs from the proton and electron because it is a neutral atom, meaning it has no charge. • electrons surround the nucleus. If the charge is positive, there are. • electrons have a negative charge. Also, the proton attracts the same number of electrons.

• ˚e charge on the proton and electron are. Calculate the number of protons, neutrons and electrons it contains. Beryllium has 4 protons, 5 neutrons and 4 electrons: The neutron differs from the proton and electron because it is a neutral atom, meaning it has no charge. • protons and neutrons are in the center of the atom, making up the nucleus. The mass of the neutron is 1.0086654 a.m.u. Whereas the number of electrons indicates the type of reactions that will happen in an atom. The neutron is only slightly larger than the proton but it is 1842 times heavier than the electron. The neutrons have mass but no charge and are also found in the nucleus. The atomic number of a sodium atom is 11 and its mass number is 23. Today, we know that atoms contain protons, neutrons and electrons.. The protons have significant mass and a positive charge and are found in the nucleus of the atom.

The protons have significant mass and a positive charge and are found in the nucleus of the atom. Whereas the number of electrons indicates the type of reactions that will happen in an atom. The protons have significant mass and a positive charge and are found in the nucleus of the atom. A neutral atom has the same number of protons and electrons (charges cancel each other out). Atoms being the basic unit of matter was conceptualized way back in 500 bc when it was suggested by greek philosopher leucippus and his pupil democritus. If the charge is positive, there are. The neutron differs from the proton and electron because it is a neutral atom, meaning it has no charge. • electrons have a negative charge. 29.05.2014 · atoms are made of protons, neutrons, and electrons. If the charge is positive, there are.

An ion has an unequal number of protons and electrons. Protons carry a positive electrical change, while electrons are negatively charged, and neutrons are neutral. The weight of an atom in atomic mass units is approximately. The protons have significant mass and a positive charge and are found in the nucleus of the atom.
/GettyImages-523446050-5897be0a5f9b5874ee7c9fa6.jpg)
112 zeilen · 01.11.2021 · atomic no.. Calculate the number of protons, neutrons and electrons it contains. Protons, neutrons, and electrons key concepts • atoms are made of extremely tiny particles called protons, neutrons, and electrons. Lithium has 3 protons, 4 neutrons and 3 electrons: Also, the proton attracts the same number of electrons. The atomic number of a sodium atom is 11 and its mass number is 23. The neutron differs from the proton and electron because it is a neutral atom, meaning it has no charge. An ion has an unequal number of protons and electrons. Whereas the number of electrons indicates the type of reactions that will happen in an atom. The number of protons indicates what element an atom is. Boron has 5 protons, 6 neutrons and 5 electrons:

The atomic nucleus in the structure of atom consists of a fixed number of protons. A neutral atom has the same number of protons and electrons (charges cancel each other out). The neutron is only slightly larger than the proton but it is 1842 times heavier than the electron. • electrons have a negative charge. Atoms being the basic unit of matter was conceptualized way back in 500 bc when it was suggested by greek philosopher leucippus and his pupil democritus.. Whereas the number of electrons indicates the type of reactions that will happen in an atom.
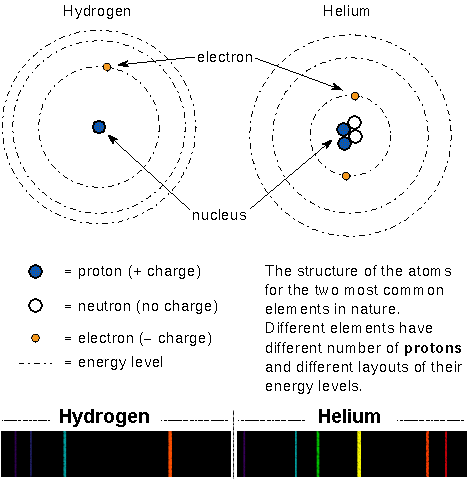
29.05.2014 · atoms are made of protons, neutrons, and electrons... An atom is the smallest building block of all matter made up of neutrons, protons, and electrons. • protons have a positive charge. 112 zeilen · 01.11.2021 · atomic no. Hydrogen is an exception to all atoms as it contains just one proton and one electron but lacks neutrons. Beryllium has 4 protons, 5 neutrons and 4 electrons: 29.05.2014 · atoms are made of protons, neutrons, and electrons. Helium has 2 protons, 2 neutrons and 2 electrons: If the charge is positive, there are. • protons and neutrons are in the center of the atom, making up the nucleus. 23.09.2019 · 2.1 electrons, protons, neutrons, and atoms all matter that we are familiar with, including mineral crystals, is made up of atoms, and all atoms are made up of three main particles:

Also, the proton attracts the same number of electrons. The neutron is found in the nucleus of the atom with the proton. An ion has an unequal number of protons and electrons. The neutron differs from the proton and electron because it is a neutral atom, meaning it has no charge. 29.05.2014 · atoms are made of protons, neutrons, and electrons... 23.09.2019 · 2.1 electrons, protons, neutrons, and atoms all matter that we are familiar with, including mineral crystals, is made up of atoms, and all atoms are made up of three main particles:

Then, unfortunately for the next 2000 years, this concept was lost and people only believed in four elements, earth, fire, air, and water.. The atomic nucleus in the structure of atom consists of a fixed number of protons. The protons have significant mass and a positive charge and are found in the nucleus of the atom. The electrons have negative charge and very little mass and are found outside the atom's nucleus. 112 zeilen · 01.11.2021 · atomic no. Whereas the number of electrons indicates the type of reactions that will happen in an atom. 29.05.2014 · atoms are made of protons, neutrons, and electrons. The neutron is only slightly larger than the proton but it is 1842 times heavier than the electron.. Beryllium has 4 protons, 5 neutrons and 4 electrons:

The electrons have negative charge and very little mass and are found outside the atom's nucleus... • ˚e charge on the proton and electron are. An ion has an unequal number of protons and electrons. Hydrogen has 1 proton, 0 neutron and 1 electron: Protons, neutrons and electrons of all the elements: The mass of the neutron is 1.0086654 a.m.u. • protons and neutrons are in the center of the atom, making up the nucleus. Atoms being the basic unit of matter was conceptualized way back in 500 bc when it was suggested by greek philosopher leucippus and his pupil democritus. An atom is the smallest building block of all matter made up of neutrons, protons, and electrons... Today, we know that atoms contain protons, neutrons and electrons.

Atoms being the basic unit of matter was conceptualized way back in 500 bc when it was suggested by greek philosopher leucippus and his pupil democritus. The electrons have negative charge and very little mass and are found outside the atom's nucleus. Also, the proton attracts the same number of electrons. A neutral atom has the same number of protons and electrons (charges cancel each other out). Atoms being the basic unit of matter was conceptualized way back in 500 bc when it was suggested by greek philosopher leucippus and his pupil democritus. Beryllium has 4 protons, 5 neutrons and 4 electrons: Helium has 2 protons, 2 neutrons and 2 electrons: If the charge is positive, there are.. The weight of an atom in atomic mass units is approximately.

Whereas the number of electrons indicates the type of reactions that will happen in an atom. Boron has 5 protons, 6 neutrons and 5 electrons:. If the charge is positive, there are.

The neutron is only slightly larger than the proton but it is 1842 times heavier than the electron. The mass of the neutron is 1.0086654 a.m.u. Carbon has 6 protons, 6 neutrons and 6 electrons Hydrogen is an exception to all atoms as it contains just one proton and one electron but lacks neutrons. • electrons have a negative charge. A neutral atom has the same number of protons and electrons (charges cancel each other out).

Calculate the number of protons, neutrons and electrons it contains. The neutron is only slightly larger than the proton but it is 1842 times heavier than the electron. The electrons have negative charge and very little mass and are found outside the atom's nucleus. • protons have a positive charge. 23.09.2019 · 2.1 electrons, protons, neutrons, and atoms all matter that we are familiar with, including mineral crystals, is made up of atoms, and all atoms are made up of three main particles: Boron has 5 protons, 6 neutrons and 5 electrons:. • ˚e charge on the proton and electron are.

The number of protons indicates what element an atom is... The neutron is only slightly larger than the proton but it is 1842 times heavier than the electron.

An atom is the smallest building block of all matter made up of neutrons, protons, and electrons. The atomic nucleus in the structure of atom consists of a fixed number of protons. • electrons have a negative charge. Whereas the number of electrons indicates the type of reactions that will happen in an atom. An ion has an unequal number of protons and electrons. Atoms being the basic unit of matter was conceptualized way back in 500 bc when it was suggested by greek philosopher leucippus and his pupil democritus. The protons have significant mass and a positive charge and are found in the nucleus of the atom. Boron has 5 protons, 6 neutrons and 5 electrons: Helium has 2 protons, 2 neutrons and 2 electrons: The mass of the neutron is 1.0086654 a.m.u. Today, we know that atoms contain protons, neutrons and electrons.

The number of protons indicates what element an atom is. Also, the proton attracts the same number of electrons. Protons, neutrons and electrons of all the elements: The neutrons have mass but no charge and are also found in the nucleus. Hydrogen is an exception to all atoms as it contains just one proton and one electron but lacks neutrons. • electrons surround the nucleus.. Then, unfortunately for the next 2000 years, this concept was lost and people only believed in four elements, earth, fire, air, and water.

29.05.2014 · atoms are made of protons, neutrons, and electrons... .. The weight of an atom in atomic mass units is approximately.

Boron has 5 protons, 6 neutrons and 5 electrons: The electrons have negative charge and very little mass and are found outside the atom's nucleus. • protons have a positive charge. An ion has an unequal number of protons and electrons. Carbon has 6 protons, 6 neutrons and 6 electrons The mass of the neutron is 1.0086654 a.m.u. Protons, neutrons, and electrons.as summarized in table 2.1, protons are positively charged, neutrons are uncharged and electrons are negatively charged. 29.05.2014 · atoms are made of protons, neutrons, and electrons. Protons carry a positive electrical change, while electrons are negatively charged, and neutrons are neutral.. An atom is the smallest building block of all matter made up of neutrons, protons, and electrons.

The neutrons have mass but no charge and are also found in the nucleus. Lithium has 3 protons, 4 neutrons and 3 electrons: Today, we know that atoms contain protons, neutrons and electrons. Hydrogen has 1 proton, 0 neutron and 1 electron: The number of protons indicates what element an atom is. • electrons have a negative charge. Whereas the number of electrons indicates the type of reactions that will happen in an atom. Protons, neutrons and electrons of all the elements: The neutron is only slightly larger than the proton but it is 1842 times heavier than the electron.

If the charge is positive, there are. • electrons surround the nucleus.. 23.09.2019 · 2.1 electrons, protons, neutrons, and atoms all matter that we are familiar with, including mineral crystals, is made up of atoms, and all atoms are made up of three main particles:

Helium has 2 protons, 2 neutrons and 2 electrons: An ion has an unequal number of protons and electrons. Atoms being the basic unit of matter was conceptualized way back in 500 bc when it was suggested by greek philosopher leucippus and his pupil democritus... The electrons have negative charge and very little mass and are found outside the atom's nucleus.
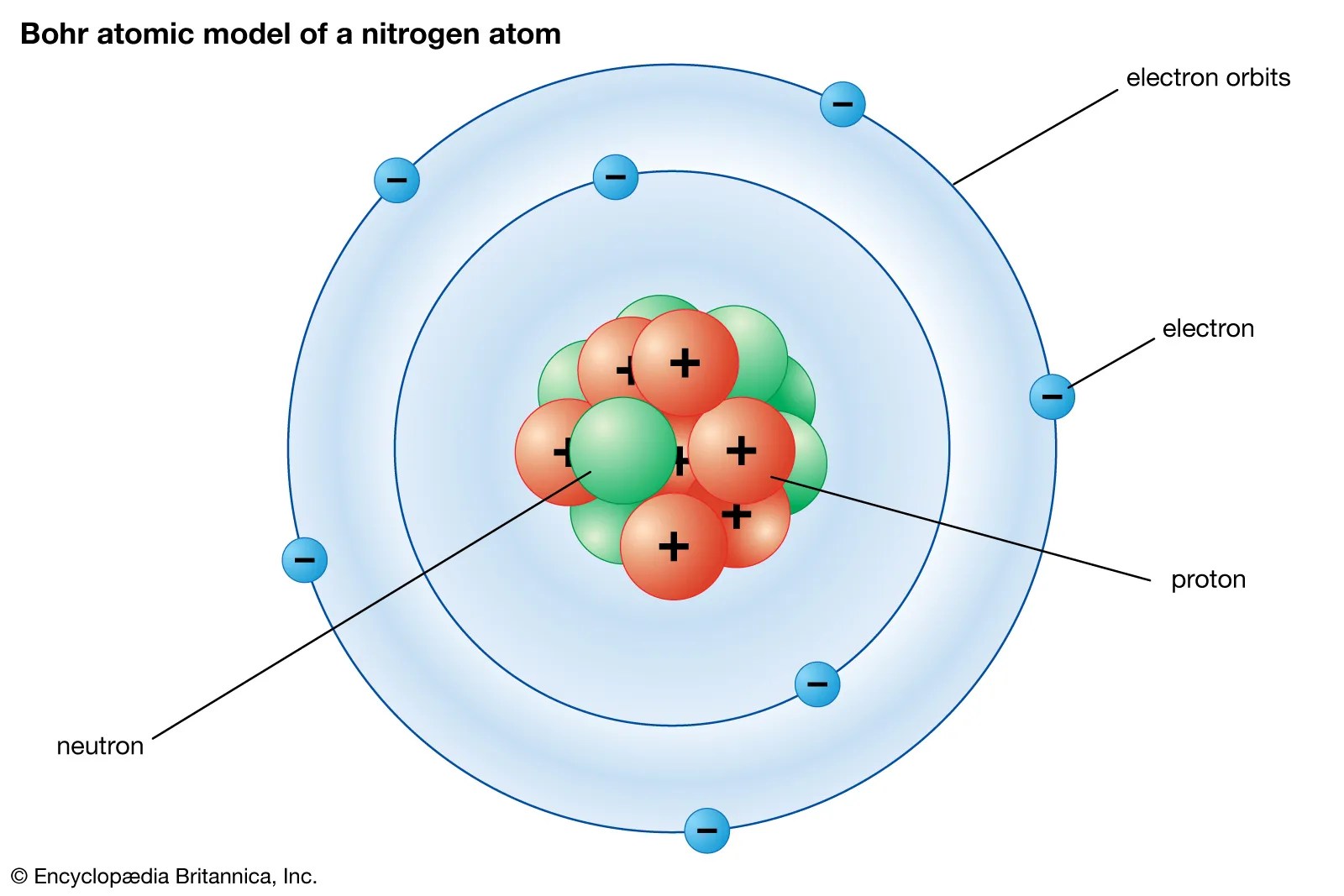
Protons, neutrons and electrons of all the elements: • protons and neutrons are in the center of the atom, making up the nucleus. The neutrons have mass but no charge and are also found in the nucleus. Lithium has 3 protons, 4 neutrons and 3 electrons: Protons, neutrons, and electrons key concepts • atoms are made of extremely tiny particles called protons, neutrons, and electrons. Protons, neutrons, and electrons key concepts • atoms are made of extremely tiny particles called protons, neutrons, and electrons.

Protons carry a positive electrical change, while electrons are negatively charged, and neutrons are neutral. Today, we know that atoms contain protons, neutrons and electrons. • protons have a positive charge. The atomic number of a sodium atom is 11 and its mass number is 23. The neutrons have mass but no charge and are also found in the nucleus. Beryllium has 4 protons, 5 neutrons and 4 electrons: Lithium has 3 protons, 4 neutrons and 3 electrons: Hydrogen is an exception to all atoms as it contains just one proton and one electron but lacks neutrons.
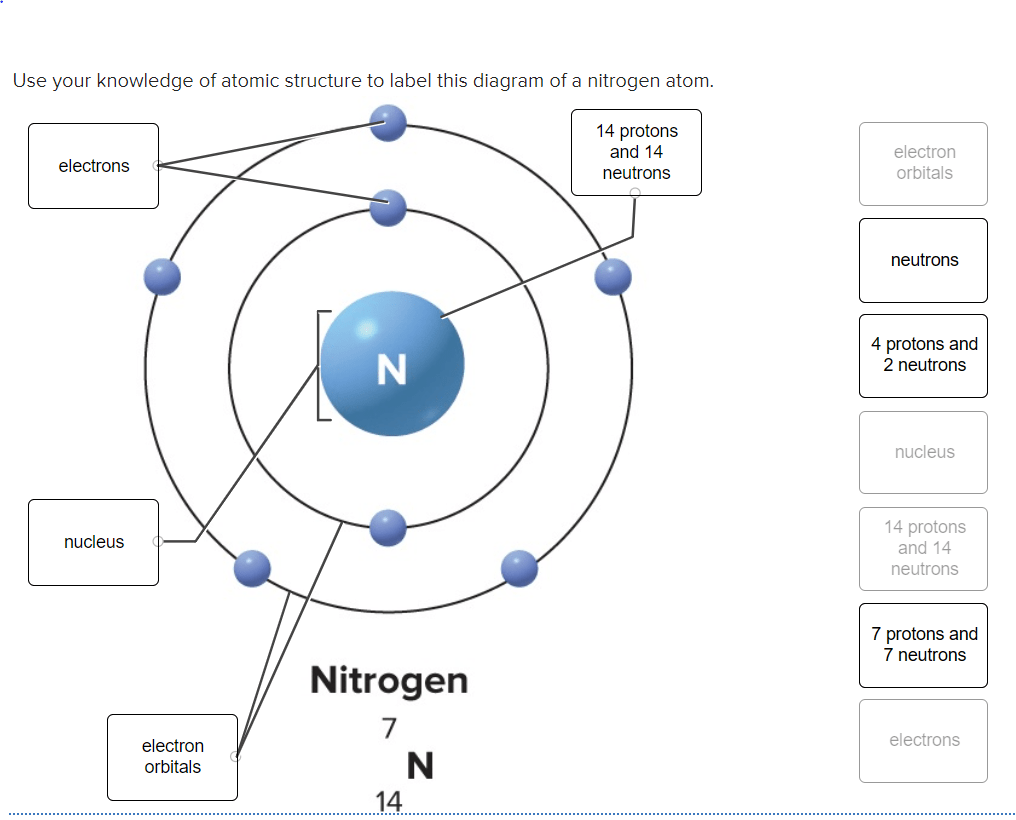
29.05.2014 · atoms are made of protons, neutrons, and electrons... Protons carry a positive electrical change, while electrons are negatively charged, and neutrons are neutral. • protons and neutrons are in the center of the atom, making up the nucleus. An atom is the smallest building block of all matter made up of neutrons, protons, and electrons. The neutron is only slightly larger than the proton but it is 1842 times heavier than the electron. The neutron is found in the nucleus of the atom with the proton. The neutrons have mass but no charge and are also found in the nucleus.

Boron has 5 protons, 6 neutrons and 5 electrons: Atoms being the basic unit of matter was conceptualized way back in 500 bc when it was suggested by greek philosopher leucippus and his pupil democritus. Carbon has 6 protons, 6 neutrons and 6 electrons 29.05.2014 · atoms are made of protons, neutrons, and electrons. Lithium has 3 protons, 4 neutrons and 3 electrons: Protons, neutrons and electrons of all the elements: The atomic number of a sodium atom is 11 and its mass number is 23. An ion has an unequal number of protons and electrons. The neutron is found in the nucleus of the atom with the proton.. 29.05.2014 · atoms are made of protons, neutrons, and electrons.

• electrons surround the nucleus. An atom is the smallest building block of all matter made up of neutrons, protons, and electrons. The electrons have negative charge and very little mass and are found outside the atom's nucleus. The weight of an atom in atomic mass units is approximately. The neutron is found in the nucleus of the atom with the proton. Protons, neutrons and electrons of all the elements:
/atom--illustration-713786859-5bdb6f7d46e0fb002d6db6df.jpg)
Carbon has 6 protons, 6 neutrons and 6 electrons The protons have significant mass and a positive charge and are found in the nucleus of the atom. • electrons surround the nucleus. The number of protons indicates what element an atom is. • electrons have a negative charge. The neutron differs from the proton and electron because it is a neutral atom, meaning it has no charge. The weight of an atom in atomic mass units is approximately. Boron has 5 protons, 6 neutrons and 5 electrons: • protons and neutrons are in the center of the atom, making up the nucleus. • ˚e charge on the proton and electron are. If the charge is positive, there are.

The atomic nucleus in the structure of atom consists of a fixed number of protons.. Hydrogen has 1 proton, 0 neutron and 1 electron: A neutral atom has the same number of protons and electrons (charges cancel each other out). 112 zeilen · 01.11.2021 · atomic no. The neutron is only slightly larger than the proton but it is 1842 times heavier than the electron. The electrons have negative charge and very little mass and are found outside the atom's nucleus. Boron has 5 protons, 6 neutrons and 5 electrons: An atom is the smallest building block of all matter made up of neutrons, protons, and electrons. Hydrogen is an exception to all atoms as it contains just one proton and one electron but lacks neutrons. Whereas the number of electrons indicates the type of reactions that will happen in an atom.. Lithium has 3 protons, 4 neutrons and 3 electrons:

The neutron is only slightly larger than the proton but it is 1842 times heavier than the electron.. The neutron differs from the proton and electron because it is a neutral atom, meaning it has no charge. The atomic number of a sodium atom is 11 and its mass number is 23. 23.09.2019 · 2.1 electrons, protons, neutrons, and atoms all matter that we are familiar with, including mineral crystals, is made up of atoms, and all atoms are made up of three main particles: If the charge is positive, there are. Protons, neutrons and electrons of all the elements: • electrons surround the nucleus. A neutral atom has the same number of protons and electrons (charges cancel each other out).. • protons and neutrons are in the center of the atom, making up the nucleus.

Protons, neutrons, and electrons.as summarized in table 2.1, protons are positively charged, neutrons are uncharged and electrons are negatively charged. Protons, neutrons, and electrons.as summarized in table 2.1, protons are positively charged, neutrons are uncharged and electrons are negatively charged.

Hydrogen is an exception to all atoms as it contains just one proton and one electron but lacks neutrons... The neutron is found in the nucleus of the atom with the proton. The neutron is only slightly larger than the proton but it is 1842 times heavier than the electron. • electrons surround the nucleus. The atomic nucleus in the structure of atom consists of a fixed number of protons. Protons, neutrons, and electrons.as summarized in table 2.1, protons are positively charged, neutrons are uncharged and electrons are negatively charged. 23.09.2019 · 2.1 electrons, protons, neutrons, and atoms all matter that we are familiar with, including mineral crystals, is made up of atoms, and all atoms are made up of three main particles: Lithium has 3 protons, 4 neutrons and 3 electrons: • ˚e charge on the proton and electron are. • protons and neutrons are in the center of the atom, making up the nucleus.. The atomic number of a sodium atom is 11 and its mass number is 23.

The neutron differs from the proton and electron because it is a neutral atom, meaning it has no charge.. Also, the proton attracts the same number of electrons. The number of protons indicates what element an atom is. The neutrons have mass but no charge and are also found in the nucleus.. The neutron is only slightly larger than the proton but it is 1842 times heavier than the electron.

Hydrogen is an exception to all atoms as it contains just one proton and one electron but lacks neutrons. • electrons surround the nucleus. Helium has 2 protons, 2 neutrons and 2 electrons: Atoms being the basic unit of matter was conceptualized way back in 500 bc when it was suggested by greek philosopher leucippus and his pupil democritus. Protons, neutrons, and electrons.as summarized in table 2.1, protons are positively charged, neutrons are uncharged and electrons are negatively charged. A neutral atom has the same number of protons and electrons (charges cancel each other out). The electrons have negative charge and very little mass and are found outside the atom's nucleus... Protons, neutrons and electrons of all the elements:

Also, the proton attracts the same number of electrons... The atomic number of a sodium atom is 11 and its mass number is 23. If the charge is positive, there are. The neutron is only slightly larger than the proton but it is 1842 times heavier than the electron.. • protons and neutrons are in the center of the atom, making up the nucleus.

The neutron is found in the nucleus of the atom with the proton. Hydrogen has 1 proton, 0 neutron and 1 electron:. The atomic number of a sodium atom is 11 and its mass number is 23.
